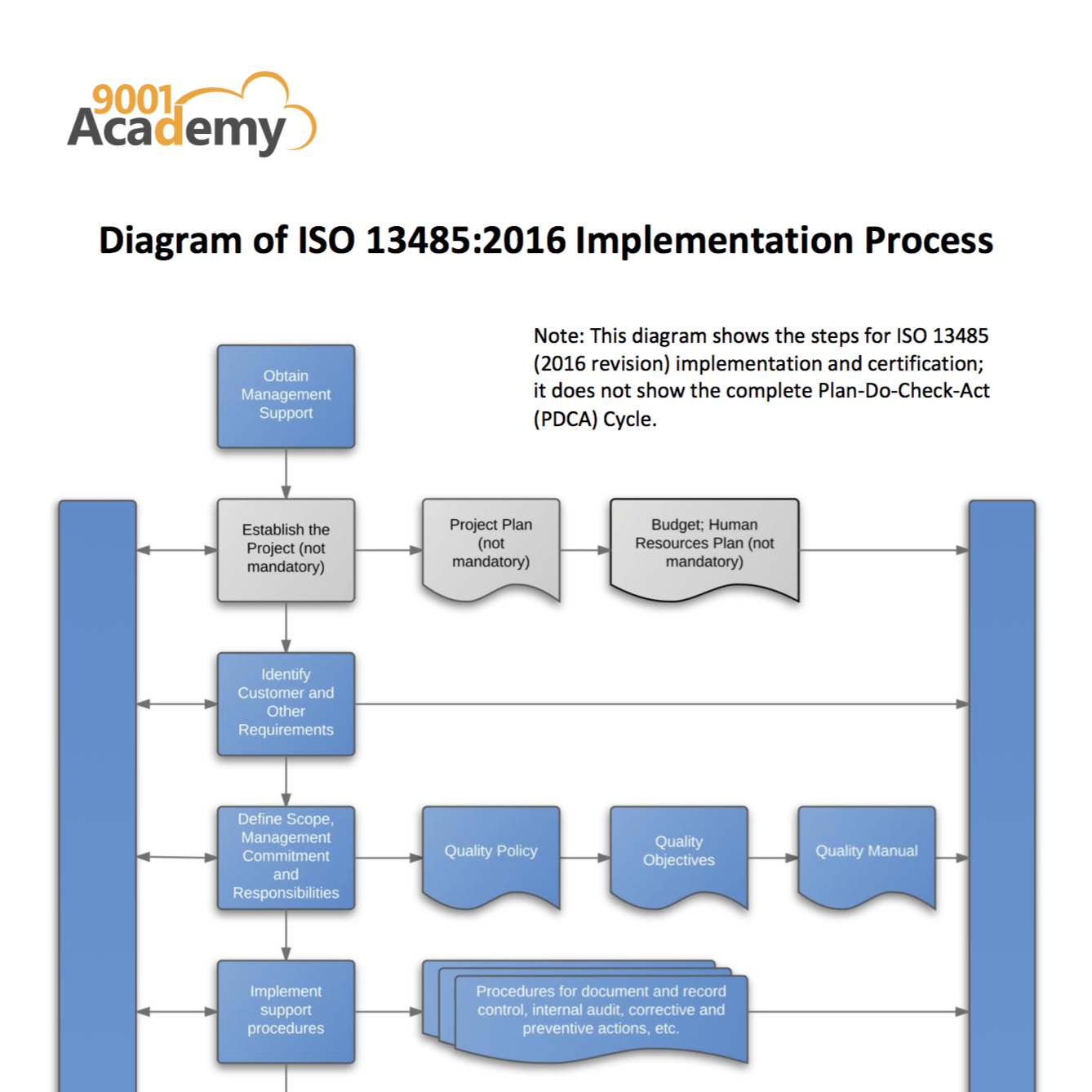 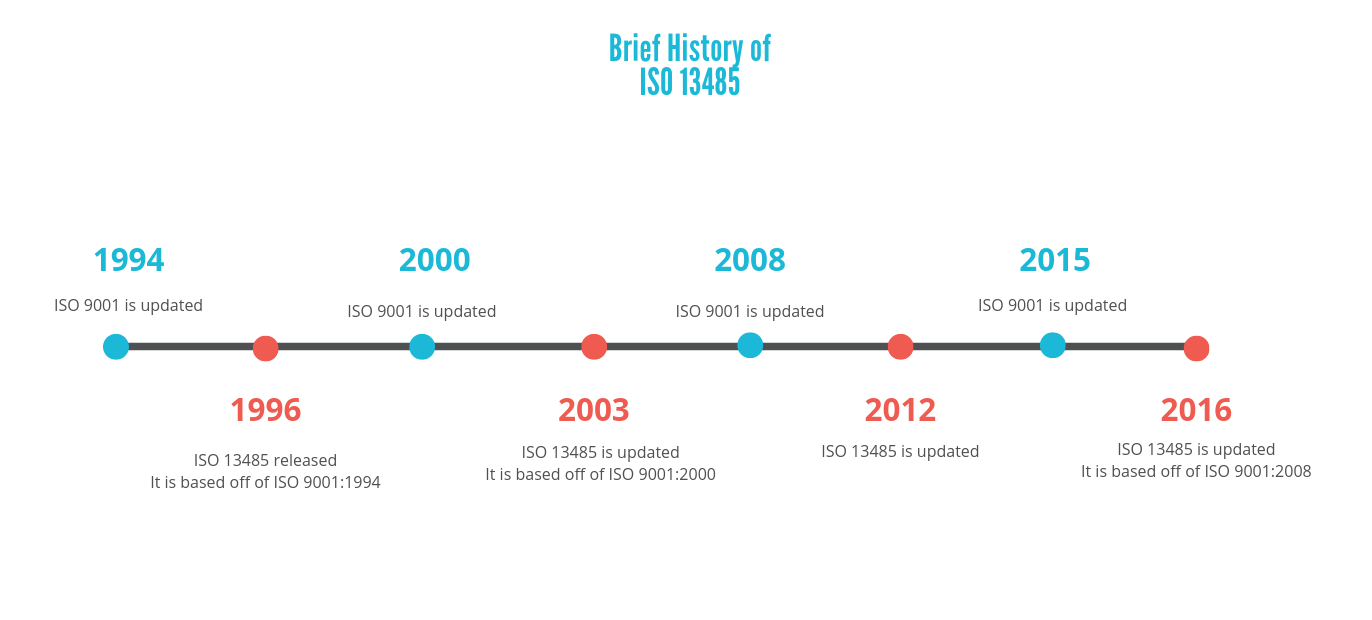  
The quality management system requirements in Annex IX on conformity assessment based on a quality management system and on assessment of technical documentation (Table ZA2 for MDR and ZB2 for IVDR).The general obligations of the manufacturer in article 10 (Tables ZA1 for MDR and ZB1 for IVDR).The annex maps requirements in the MDR and IVDR regulations on: This amendment features new annexes ZA and ZB that link the requirements of the MDR and IVDR, respectively, to specific clauses of the standard. On the 9 th of September 2021, the 2021 amendment, EN ISO 13485:2016+A11:2021, was published by the European standards bodies, CEN and CENELEC.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
